The mirror generates a virtual image of the flower that appears to be behind the mirror. 
Mass of unit cell = 4 atoms * 3.19 x 10^-22 g/atom ≈ 1.Trending Questions What happens to energy when it's transformed and transferred? What is a heavy wind called? Why the absence of motion is called absolute zero? What wave has a color spectrum that is known as roygbiv? How do glow sticks light up? How does the sun produce energy from matter in its core through the process of? Why as water is bad conductor of electricity is it used to generate hydro electric power? What is loading efficiency? Where you will you encounter a higher pitched sound? How You place a flower 18 feet from a concave mirror. Now, we can find the mass of the atoms in the unit cell: Therefore, the mass of one iridium atom is: Calculate the radius of an iridium atom, given that Ir has an FCC crystal structure, a density of 22.4 g/cm3, and an atomic weight of 192.2 g/mol. The molar mass of iridium is 192.22 g/mol. The mass of one iridium atom can be found using the molar mass of iridium metal and Avogadros number: Mass of one iridium atom (Molar mass of iridium)/(Avogadros number) (192.22 g/mol)/(6.022 x 1023 atoms/mol) 3. A very hard, brittle, silvery-white transition metal of the platinum group, it is considered the second-densest naturally occurring metal (after osmium) with a density of 22.56 g/cm 3 (0.815 lb/cu in) 6 as defined by experimental X-ray crystallography. In a face-centered cubic unit cell, there are four iridium atoms. In a face-centered cubic unit cell, there are 4 atoms per unit cell (1/8 of an atom at each of the 8 corners and 1/2 of an atom at each of the 6 faces). Density is defined as mass per unit volume. (b) To calculate the density of iridium metal, we need to find the mass of the atoms in the unit cell and divide it by the volume of the unit cell. So, the atomic radius of an iridium atom is approximately 1.36 Å. Now, we can plug in the given edge length (a = 3.833 Å) and solve for the atomic radius (r): Since this distance is equal to the sum of the radii of the two atoms, and both atoms are iridium, we can write: 
Now, we can find the distance between the center of the face atom and the corner atom: Let's denote the edge length as a and the face diagonal as d: 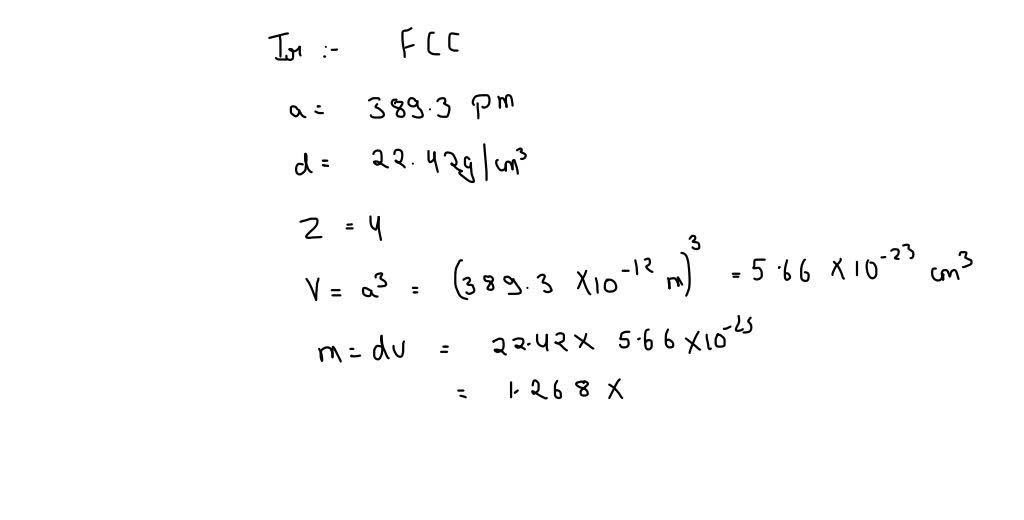
We can use the Pythagorean theorem to find the length of the face diagonal. Therefore, the distance between the center of the face atom and the corner atom is half of the face diagonal. Express your answer using four significant figures. The edge length will help us to determine the volume of a unit cell. Overall, a face-centered cubic structure contains four iridium atoms. Calculate the atomic radius of an iridium atoms. Iridium metal crystallizes in a face-centered cubic structure which means it contains 1/8 of an atom at each of the 8 corners and 1/2 of an atom at each of the six faces. The atom in the center of the face is in contact with the corner atoms, as shown in the drawing. In a face-centered cubic unit cell, the diagonal of the face can be divided into two equal parts by the face atom. Iridium crystallizes in a face-centered cubic unit cell that has an edge length of 3.833 A. Since the face atom is in contact with the corner atom, this distance is equal to the sum of the radii of the two atoms. Problem 11: Platinum has a density of 21.45 g/cm 3 and a unit cell side length 'd' of 3.93 ngstroms. Use these data to calculate a value for Avogadro's Number. In a face-centered cubic unit cell, the diagonal of the face can be divided into two equal parts by the face atom. Problem 10: Iridium has a face centered cubic unit cell with an edge length of 383.3 pm. Given that the length of the edge of a unit cell is 383 pm, determine the density of. Since the face atom is in contact with the corner atom, this distance is equal to the sum of the radii of the two atoms. metal iridium (Ir) crystallizes with a face-centered cubic unit cell.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |



 RSS Feed
RSS Feed
